Stran trenutno ni na voljo v vašem jeziku. Lahko si ogledate strojni prevod, ustvarjen s storitvijo Google Translate. Te storitve ne zagotavljamo mi in rezultatov prevoda nismo preverili.
Za dodatno pomoč se lahko obrnete na nas.
Pharmaceuticals
From drug discovery to formulation, development, and trouble shooting, our systems give you answers.
APIs, excipients and more
Renishaw's Raman systems have the performance and flexibility you need to analyse all your pharmaceutical materials.
- Identify and differentiate API polymorphs and excipients
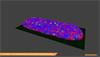
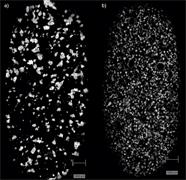
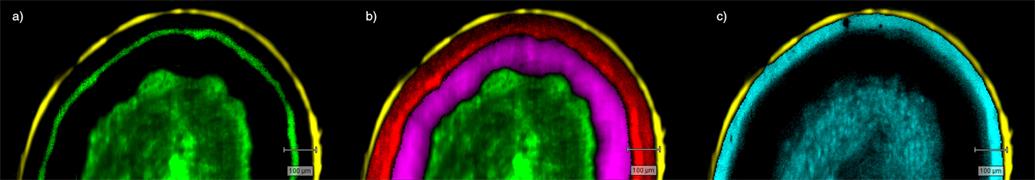
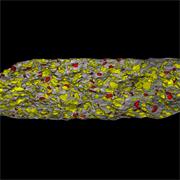
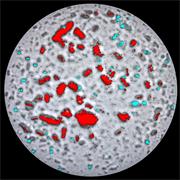
- Generate images of the formulations used in tablets, inhalers and nasal sprays at sub-micrometre spatial resolution
- Use our dedicated high-throughput polymorph screening capabilities
- Study the thermal behaviour of drugs using advanced temperature control
- Fast, quantitative analyses of entire tablets and powder blends
- Determine the uniformity of mixing
- Analyse API and metabolites in cells and tissue
- Identify contaminants
One spectrum reveals all
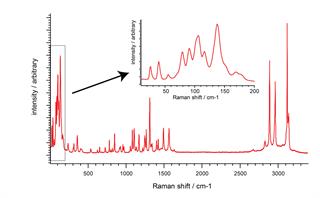
Save time. Use SynchroScan to reveal all the key Raman bands; at high spectral resolution, over the entire Raman range, in just one data collection. See lattice modes, OH bands, and everything in-between. Spot the most subtle sample differences wherever they occur in the spectrum.
Generate images rapidly
Rapidly generate images of your formulations with StreamLine™. This uses line focus illumination, allowing you to use higher laser powers without risk of sample damage, thereby reducing experiment times.
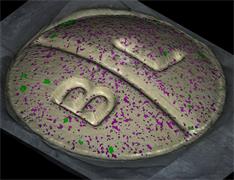
With the RA802 pharmaceutical analyser you can achieve imaging speeds of over 1500 spectra/s. It takes less than 2 minutes, from placing a standard tablet in the system, to generate a full, high resolution Raman image.
Generate images of formulations
Ensure your chemical images are representative; use Renishaw's StreamLine. You can change resolution to suit your domain size and, because Renishaw's WiRE software can cope with massive data files, you can analyse over the entire sample surface. Powerful Renishaw features, such as Slalom (to ensure the whole surface is sampled) and HD imaging (to get crisp clear images), provide all the options you need, whatever your formulation.
21 CFR 11
Renishaw's WiRE software supports 21 CFR part 11 compliance.
Product choices
RA802 Pharmaceutical Analyser
inVia™ confocal Raman microscope
Renishaw's RA802 system is designed exclusively for the pharmaceutical industry. The RA802 is a chemical imaging system optimised for routine analysis with the speed, automation and precision you need for reliable results. It is ideal for use in busy laboratories where the rapid analysis of multiple samples is required and space is at a premium.
Renishaw's inVia is a fully configurable research grade Raman microscope. Scientists worldwide trust inVia and its unparalleled flexibility. It can be upgraded, modified and customised, without compromising performance. Add accessories, lasers, fibre optic probes or combine with other analytical techniques. Whatever configuration you choose, you will have the most flexible Raman system on the market.
We're here when you need us
To find out more about this application area, or an application that isn't covered here, contact our applications team.
Contact our applications team
Downloads: chemical sciences (pharmaceuticals)
-
 Brochure: Solutions for pharmaceutical analysis
Brochure: Solutions for pharmaceutical analysis
Renishaw is a world leader in Raman spectroscopy instrumentation, with over 25 years’ experience supplying Raman systems to customers worldwide. We are a global company, with a worldwide network of scientists and engineers who are on-hand to provide you with expert product, applications and technical support. We understand the many challenges within pharmaceutical analysis and our flexible solutions have been designed with your needs in mind. Whether novice or expert, formulator or quality analyst, we have a system to meet your requirements.
-
 Product note: RA802 Pharmaceutical Analyser
Product note: RA802 Pharmaceutical Analyser
Designed exclusively for the pharmaceutical industry, the RA802 rapidly obtains detailed information on the distribution of chemical species.
-
 Application note: New methods for determining content uniformity of formulations
Application note: New methods for determining content uniformity of formulations
Using the RA802 to monitor the uniformity of a drug product’s contents to ensure that the correct dose is delivered every time.
-
 Application note: Study the stability of pharmaceutical formulations with Raman microscopy
Application note: Study the stability of pharmaceutical formulations with Raman microscopy
Many pharmaceutical materials are highly sensitive to changes in temperature and humidity which can affect their crystalline structure. Knowing the stability of these polymorphs and hydrates, in different conditions, is critical to ensure products are safe, efficient and can be patent protected.
-
 Application note: Investigate pharmaceutical solid dose products using the inVia confocal Raman microscope
Application note: Investigate pharmaceutical solid dose products using the inVia confocal Raman microscope
With so many components and properties, determining the composition, domain size and distribution of patent-expired commercially available formulations is highly desirable. This information creates opportunities to replicate an existing product without the need for lengthy development and prohibitive costs.
-
 Application note: Transmission Raman for monitoring pharmaceuticals
Application note: Transmission Raman for monitoring pharmaceuticals
Whether at the research and development stage, or manufacturing, the inVia Raman microscope offers simple and fast transmission Raman analysis.
-
 Application note: Combined chemical and particle distribution analysis of pharmaceutical powders
Application note: Combined chemical and particle distribution analysis of pharmaceutical powders
When formulating pharmaceuticals, it is important to understand the chemical composition and morphologies of the ingredients, as these can lead to changes in product performance. For example, active ingredient co-localisation and particle adhesion can affect the efficacy of formulation. This application note details how to use a Renishaw Raman system to conduct this type of analysis.
-
 Application note: Using Raman spectroscopy to tackle polymorphism, an industry problem
Application note: Using Raman spectroscopy to tackle polymorphism, an industry problem
This note looks at incidences of polymorphism in drug development including advantages and challenges.
-
 Application note: Raman imaging for deformulating a transdermal patch
Application note: Raman imaging for deformulating a transdermal patch
An application note detailing the RA802's ability to deformulate the make-up of a transdermal nicotine patch.
-
 Application note: Rapid polymorph identification with the RA802 Pharmaceutical Analyser
Application note: Rapid polymorph identification with the RA802 Pharmaceutical Analyser
Characterising the polymorphism of active pharmaceutical ingredients when developing a commercially viable product.
-
 Technical note: Quantitative Raman spectroscopy
Technical note: Quantitative Raman spectroscopy
This document focuses on concentration measurements but the principles detailed are also applicable to other quantitative measurements, such as stress/strain.
-
 White paper: Finding fakes – using Raman imaging to identify counterfeit medicines
White paper: Finding fakes – using Raman imaging to identify counterfeit medicines
Raman spectroscopy is highly specific and well-suited to identifying counterfeits which often use similar components to the reference product.
-
 White paper: Methods for deformulating drugs
White paper: Methods for deformulating drugs
In this white paper we look at reverse engineering existing drug products using Raman imaging. We show that the Renishaw RA802 Pharmaceutical Analyser can be used to successfully characterise innovator and generic products and facilitate deformulation activities, and can characterise the components, concentrations, particle sizes and distributions of the innovator formulation, allowing generic followers to adjust their processes in order to create equivalent products, or products which are strategically different.
-
 Technical note: Polymorph identification and qualification
Technical note: Polymorph identification and qualification
This technology note illustrates the benefits of combining dedicated analysis software with a high-performance Raman instrument and how Renishaw supplies an effective, user-friendly solution to provide complementary information to the multitude of established HTS techniques commercially available.
-
 Application note: Detecting small amounts of multiple API polymorphic forms in powder blends
Application note: Detecting small amounts of multiple API polymorphic forms in powder blends
This note shows how the RA802 Pharmaceutical Analyser can be used to analyse a range of samples, such as powder blends and tablets, to screen for the presence and form of APIs
-
 Product note: Cluster analysis and data classification
Product note: Cluster analysis and data classification
The ability to group highly similar spectral data is a key result for research areas including biology and medicine. It enables qualitative conclusions to be drawn objectively based on statistics, rather than subjectively based on an analyst’s experience.